Hemay022 is a small-molecule, irreversible inhibitor of EGFR and HER2 tyrosine kinases, classified as a Class 1 chemical new drug, targeting HER2+ and ER+ advanced breast cancer.
It has received support from the National Major Scientific and Technological Special Project for "Major New Drug Innovation."
HER2 is a member of the human epidermal growth factor receptor (EGFR) family, with positive expression observed in various cancers, including breast, gastric, ovarian, esophageal, lung, and biliary tract cancers. HER2 forms homologous dimers by itself or heterodimers with other members of the EGFR family.This dimerization leads to auto-phosphorylation and/or trans-phosphorylation of specific tyrosine kinases within its intracellular domain, thereby activating downstream cellular signaling pathways that promote tumor cell proliferation, survival, and metastasis.
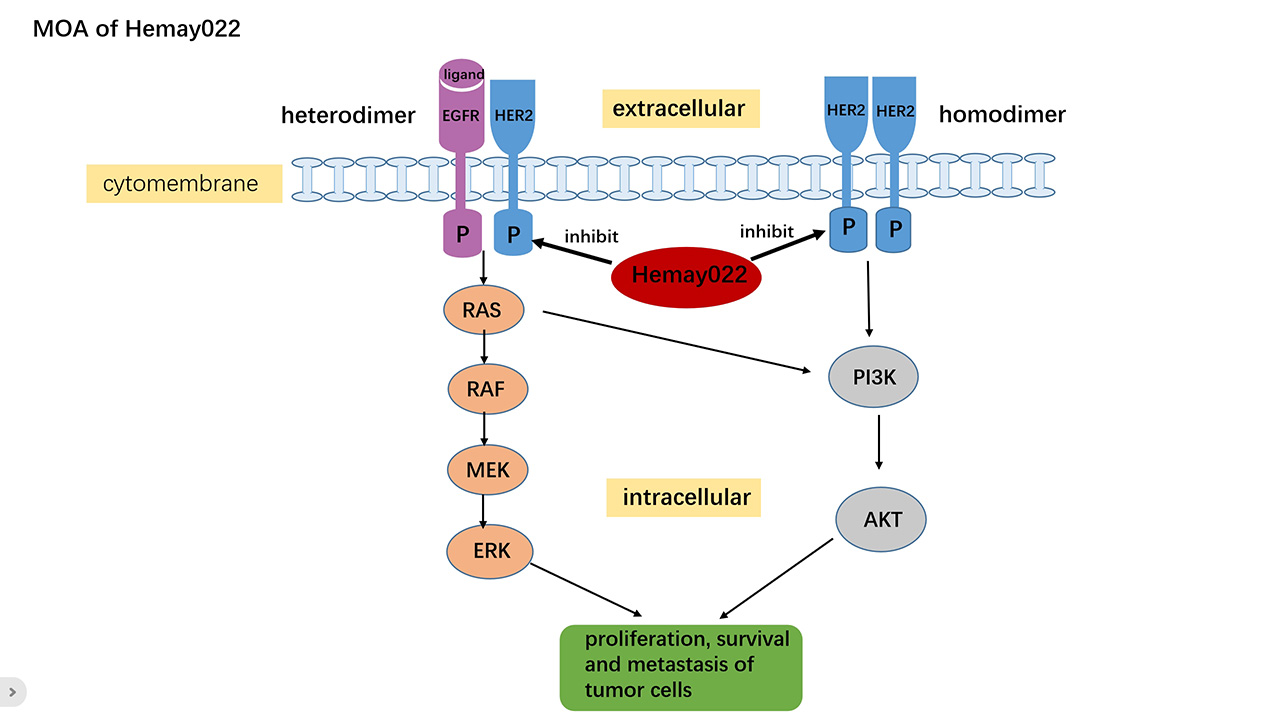
 Schematic Diagram of Hemay022's Mechanism of Action in Inhibiting Tumor Cell Growth Caused by Abnormal Activation of EGFR/HER2-Related Signaling Pathways
Schematic Diagram of Hemay022's Mechanism of Action in Inhibiting Tumor Cell Growth Caused by Abnormal Activation of EGFR/HER2-Related Signaling Pathways
Breast cancer is a prevalent malignant tumor that poses a severe threat to women's health and is the leading cause of cancer-related deaths among women. The etiology of breast cancer has not been fully elucidated; however, numerous studies indicate that its occurrence is associated with long-term stimulation by endogenous or exogenous estrogens, viruses, breast atypical hyperplasia, genetic and familial history, nutritional factors, and radiation exposure. Clinical research has revealed that the HER2 gene is amplified in 25%–30% of breast cancer patients, and its overactivation is significantly linked to the development and progression of breast cancer. HER2 has been widely recognized as a gold-standard biomarker for breast cancer detection.
Hemay022 is currently conducting Phase III clinical trials for this indication in China. For detailed information on the clinical trials, please visit CLINICALTRIALS.GOV.