Mufemilast is a novel PDE4B protein expression blocker and PDE4 inhibitor, targeting autoimmune diseases such as psoriasis, ulcerative colitis, Beh?et's disease, and psoriatic arthritis. It has received support from the national "Major New Drug Creation" science and technology major project.
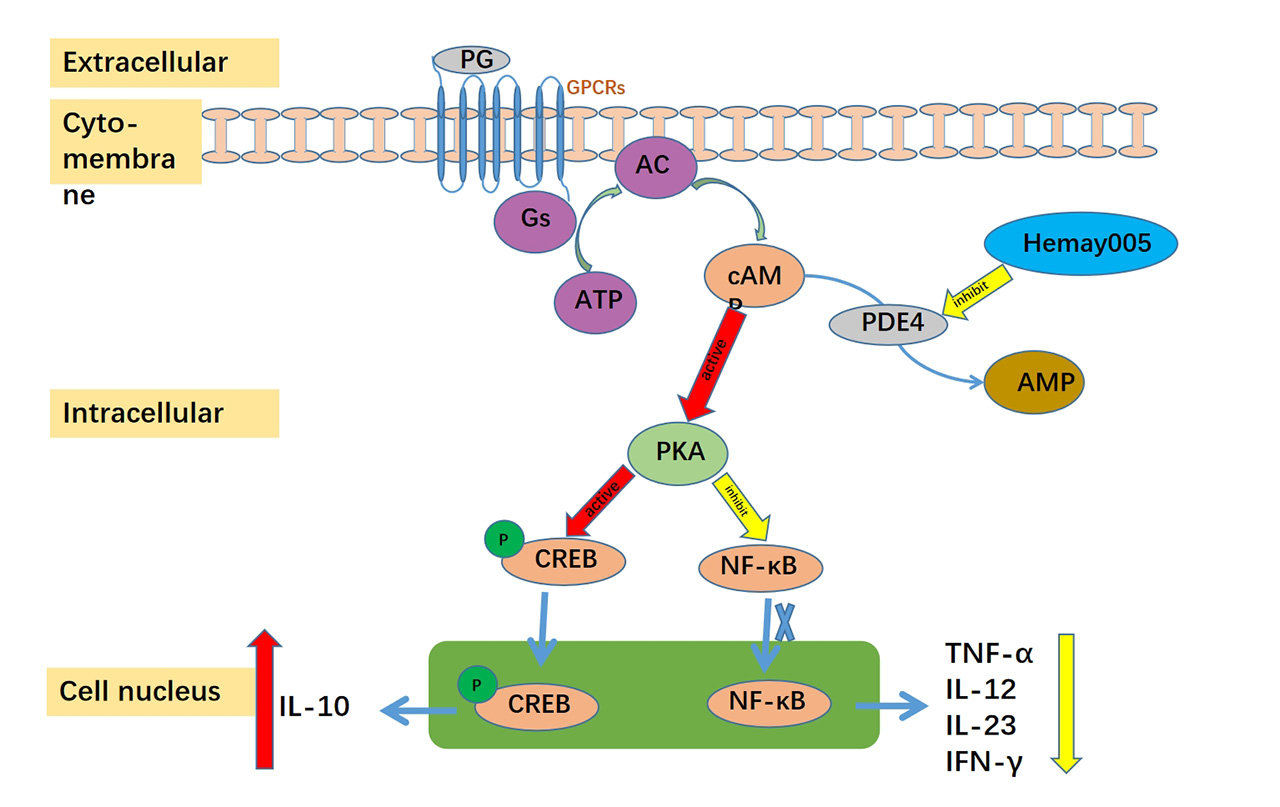
PDE4 specifically hydrolyzes cyclic adenosine monophosphate (cAMP), serving as the predominant phosphodiesterase (PDE) in mammalian inflammatory and immune cells. Given that numerous inflammatory and immune responses in the body are regulated by PDE4, selective PDE4 inhibitors can increase intracellular cAMP levels, thereby exerting broad anti-inflammatory effects.
Mufemilast reduces the hydrolysis of cAMP by blocking the over-expression of PDE4B protein and inhibiting the activity of PDE4, thus increasing the intracellular cAMP level. This effectively suppresses the proliferation of key cells (such as Th1/Th2/Th17) involved in the pathogenesis of autoimmune diseases and the production of inflammatory factors, with minimal side effects and high safety.
Psoriasis is a common chronic skin disease, colloquially known as "psoriasis vulgaris." It is not merely a dermatological condition but rather an autoimmune-inflammatory skin disease resulting from the interplay of genetic predisposition and environmental factors. The disease is characterized by abnormal immune responses that lead to the production of various inflammatory cytokines and mediators. These cytokines and mediators act on target cells, triggering aberrant cellular signal transduction. In this immune response, T cells, keratinocytes, dendritic cells, and monocytes/macrophages collaborate to drive disease pathogenesis. Due to its recalcitrance to treatment, psoriasis remains a significant research focus in the global dermatological community and is one of the priority diseases for prevention and management worldwide.
Mufemilast has obtained the "Drug Registration Certificate" for the treatment of psoriasis. For detailed information on related clinical trials, please visitCLINICALTRIALS.GOV。
Behcet's disease (BD), also known as Behcet's syndrome, was first reported by the Turkish dermatologist Behcet in 1937. The pathogenesis of BD remains unclear but may be associated with genetic factors, immune dysregulation, infections, living environment, and other factors. Abnormal levels of trace elements and heat shock proteins may also play a role in the onset of BD. Recent studies have identified susceptibility genes such as HLA-B51, TNF, IL-27, IL-23R/IL-12RB2, MMP-9, NFKB1/NFKBIA, MDR1, and PON1-L55M as being associated with BD, particularly HLA-B51, which shows a significant correlation with the disease. T cells, including Th1, Th2, Th17, γδT cells, and related cytokines such as IL-17, IL-23, IL-6, IL-8, and TNF-α, also play crucial roles in the pathogenesis of BD. The global incidence of BD varies, with higher prevalence in the Mediterranean, Middle East, and East Asia. BD predominantly affects individuals aged 15-50 years.
Mufemilast is in phase III clinical trials for this indication in China. For details of clinical trials, visit CLINICALTRIALS.GOV.
Ulcerative Colitis (UC) is a chronic, non-specific inflammatory disease of the colon and rectum with an etiology that remains not fully understood. The lesions are confined to the mucosal and submucosal layers of the large intestine. The disease predominantly affects the sigmoid colon and rectum but can also extend to the descending colon or even involve the entire colon. The course of UC is prolonged and often characterized by recurrent episodes. The etiology and pathogenesis of UC are associated with multiple factors, including genetic predisposition, antigenic stimulation, activation of the immune system, and interactions among various environmental factors, leading to chronic intestinal inflammation.
Mufemilast is currently undergoing clinical trials for this indication in China. For detailed information on related clinical trials, please visit CLINICALTRIALS.GOV.
Mufemilast is currently conducting clinical trials for this indication in China. For detailed information on related clinical trials, please visit CLINICALTRIALS.GOV.
Mufemilast is currently conducting clinical trials for this indication in China. For detailed information on related clinical trials, please visit CLINICALTRIALS.GOV.
Mufemilast is currently conducting clinical trials for this indication in China. For detailed information on related clinical trials, please visit CLINICALTRIALS.GOV.
Mufemilast is currently conducting clinical trials for this indication in China. For detailed information on related clinical trials, please visit CLINICALTRIALS.GOV。